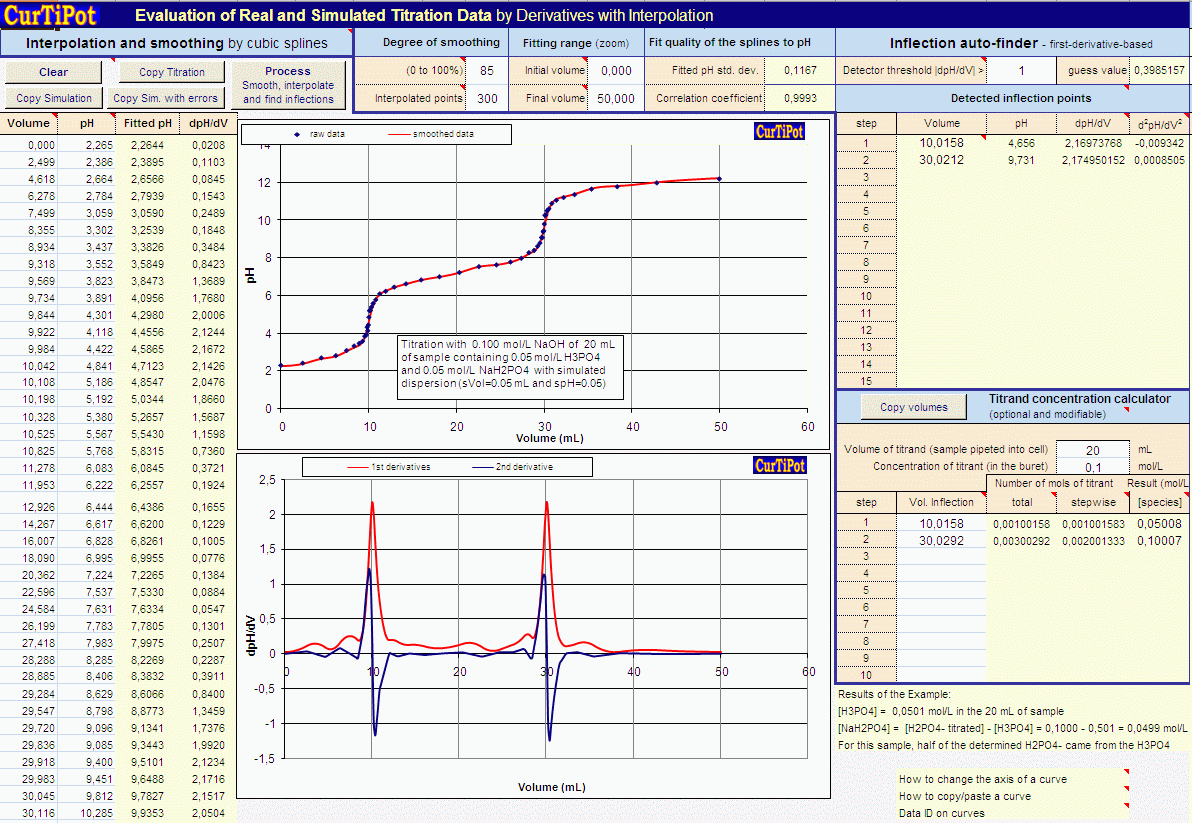
![University level Laboratory] How to calculate the calibration curve using the ATP stock? : r/HomeworkHelp University level Laboratory] How to calculate the calibration curve using the ATP stock? : r/HomeworkHelp](https://preview.redd.it/university-level-laboratory-how-to-calculate-the-v0-d85xipyq7g8a1.jpg?width=2526&format=pjpg&auto=webp&s=c80a3accbe7afb7f5f797fc1dee6b9ad91ffc97a)
University level Laboratory] How to calculate the calibration curve using the ATP stock? : r/HomeworkHelp

SciELO - Brasil - Cálculo e preparo de soluções tampão: guia completo usando o software PeakMaster<sup>®</sup> Cálculo e preparo de soluções tampão: guia completo usando o software PeakMaster<sup>®</sup>
If you have 100ml of a 0.10M TRIS buffer at pH 8.3 (pKa=8.3) and you add 3.0ml of 1 M HCl, what will be the new pH? - Quora


![Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/11/tris-base-molecular-weight-calculation-300x204.jpg)

![Tris Hydrochloride [C4H11NO3.HCl] Molecular Weight Calculation - Laboratory Notes Tris Hydrochloride [C4H11NO3.HCl] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/11/tris-hydrochloride-molecular-weight-calculation-300x225.jpg)